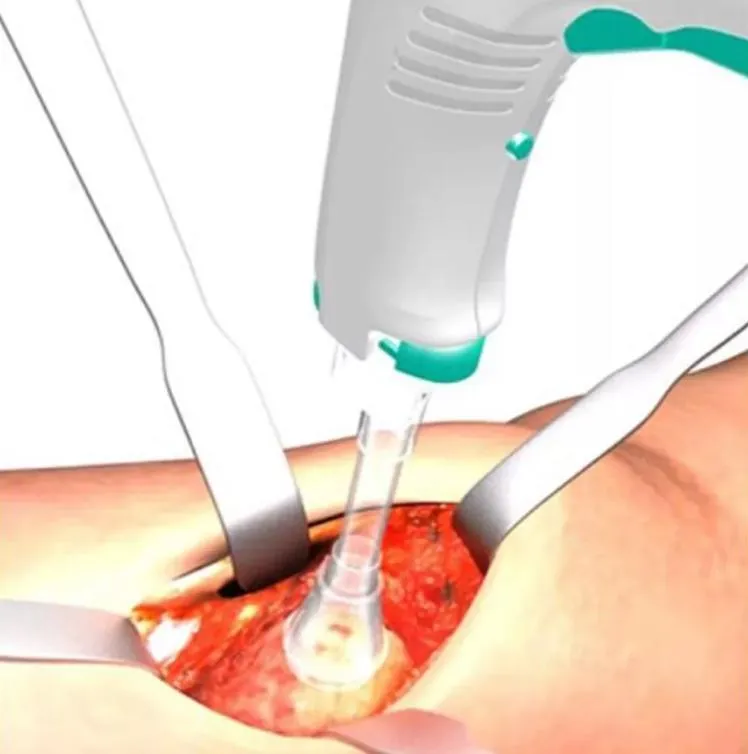
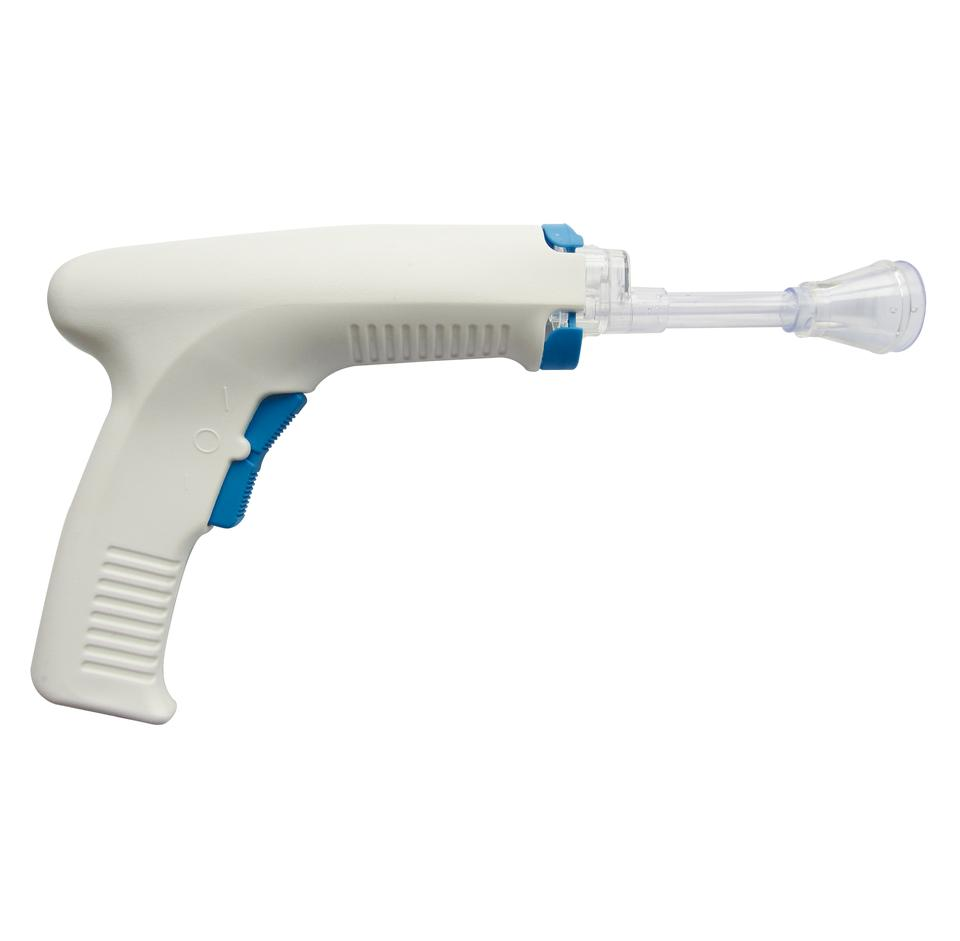
The Disposable Pulse Lavage System represents a new generation of wound cleaning technology. It is specifically designed to clean inactivated tissues, bacteria, and foreign bodies in wounds to significantly reduce the risk of infection while avoiding secondary damage to surrounding healthy tissues.
⚙️ Technical Data
Spray Distance & Flow Rate:
- High Setting: Distance ≥1000mm, Flow Rate ≥12ml/s
- Low Setting: Distance ≥500mm, Flow Rate ≥7ml/s
Spray Shapes: Umbrella Shape Head (Shower effect) and Straight Shape Head (Cylinder effect).
Sterilization: Sterilized by Epoxy methylmethane (Residual quantity < 10μg/g).
Key Features & Benefits
- Ergonomic Design: Minimal setup, easy to use, and provides a secure, comfortable grip with a textured barrel.
- Enhanced Visibility: Enlarge operation vision and shorten overall surgical time.
- Improved Outcomes: Increases permeability between bone cement and sclerotic bone.
- Risk Reduction: Reduces the incidence rate of fat embolism and the looseness rate of implants.
- Cost-Effective: Reduces the required dosage of antibiotics and overall medical expenses.
- Powerful Performance: Pressure up to 15 PSI with a flow range of 600-1200ml/min.
- Battery Powered: Powered by 7 x 1.5V AA batteries for cordless convenience.
*All the prices are only for reference. Exact price will be based on the latest quotation.
Frequently Asked Questions (FAQ)
What is the primary use of the Disposable Pulse Lavage System?
It is primarily used in orthopedic trauma and joint arthroplasty to clean inactivated tissues, bacteria, and foreign materials from wounds, effectively reducing infection risks.
How is the system powered?
The system is powered by 7 x 1.5V regular AA batteries, allowing for high portability and ease of use in surgical environments without needing external power cables.
Does the device offer different spray settings?
Yes, it features two speeds: a low speed for gentle lavage and a high speed for powerful cleaning action. It also includes different head shapes (Umbrella and Straight).
What certifications does this medical device hold?
The product is fully certified with CE and ISO13485 standards, ensuring it meets international quality and safety requirements for medical orthopedic use.
Is the product sterilized before delivery?
Yes, all units are sterilized by Epoxy methylmethane before leaving the factory, with residual quantities maintained below 10μg/g for patient safety.
What are the clinical benefits for joint surgery?
It helps increase permeability between bone cement and bone, reduces the risk of fat embolism, and decreases the post-operative rate of tissue adhesion.




 Medisage Medical
Medisage Medical