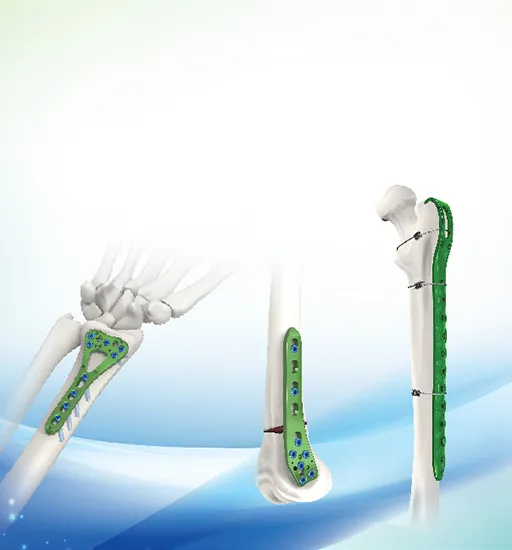
What materials are used in the Spinal System Crosslinks?
The products are manufactured using high-quality Titanium Alloy, which ensures excellent biocompatibility and mechanical strength for bone fracture fixation.
Are these implants provided sterile or non-sterile?
These are disposable medical devices. Please refer to the specific packaging instructions for sterilization status and protocols before use.
What certifications do your orthopedic products hold?
Our products are fully certified with CE and ISO13485 standards, meeting international quality requirements for medical devices.
What is the Minimum Order Quantity (MOQ)?
The MOQ for our spinal system components is as low as 1 PC, allowing for flexible procurement based on surgical needs.
Do you support OEM or ODM services?
Yes, we accept OEM and ODM requests to meet specific clinical requirements or branding needs for our global partners.
What is the typical lead time for shipping?
We utilize reliable international couriers such as DHL, UPS, FedEx, and TNT. Shipping times vary by location but are processed efficiently from our production facility.





 Medisage Medical
Medisage Medical