



 Medisage Medical
Medisage Medical










| P/N | Product Name | Size (Φ / Length) | P/N | Product Name | Size (Φ / Length) | ||
|---|---|---|---|---|---|---|---|
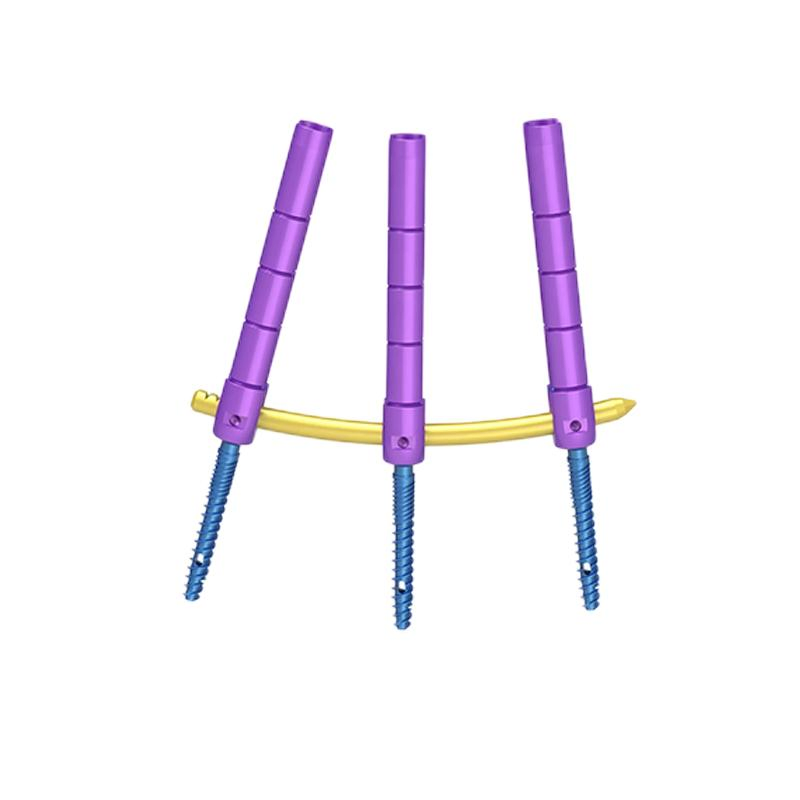
| 62601-3 | Minimally invasive monoplane screw | Φ5 | 35-45 | 62701-3 | Minimally invasive monoplane screw | Φ5 | 35-45 |
| 62604-6 | Φ5.5 | 35-45 | 62704-6 | Φ5.5 | 35-45 | ||
| 62607-9 | Φ6 | 45-50 | 62707-9 | Φ6 | 45-50 | ||
| 62610-12 | Φ6.5 | 45-50 | 62711-12 | Φ6.5 | 45-50 | ||
| 62801-17 | Straight Rod | Φ6 | 50-220 | 62901-11 | Curved Rod | Φ6 | 50-110 |


| Minimally Invasive Spine System Instrument Kit | |||||
|---|---|---|---|---|---|
| No. | Description | Specification | No. | Description | Specification |
| 63001 | Round End Guide Pin | φ1.4*490 | 63017 | Screw driver For Polyaxial Screw | - |
| 63002 | Sharp End Guide Pin | φ1.4*490 | 63018 | Ball Shape Handle | - |
| 63013 | Tap For Monoplane Screw | φ5.5 | 63028 | Rod Gauge | - |
| 63016 | Screw driver For Monoplane Screw (long) | - | 63032 | Instrument Box | 524*250*105mm |





Established in 1958, the facility is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus, the production line covers Spine pedicle screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
The products are characterized by multiple varieties and complete specifications. Advanced manufacturing equipment including imported CNC high precision digital control machines and a 100,000 Grade axenic purification chamber contribute to superior product quality. The facility was among the first to pass ISO 9001 quality system certification, with products receiving multiple national silver awards and invention prizes.


