




1 / 5
 Medisage Medical
Medisage Medical













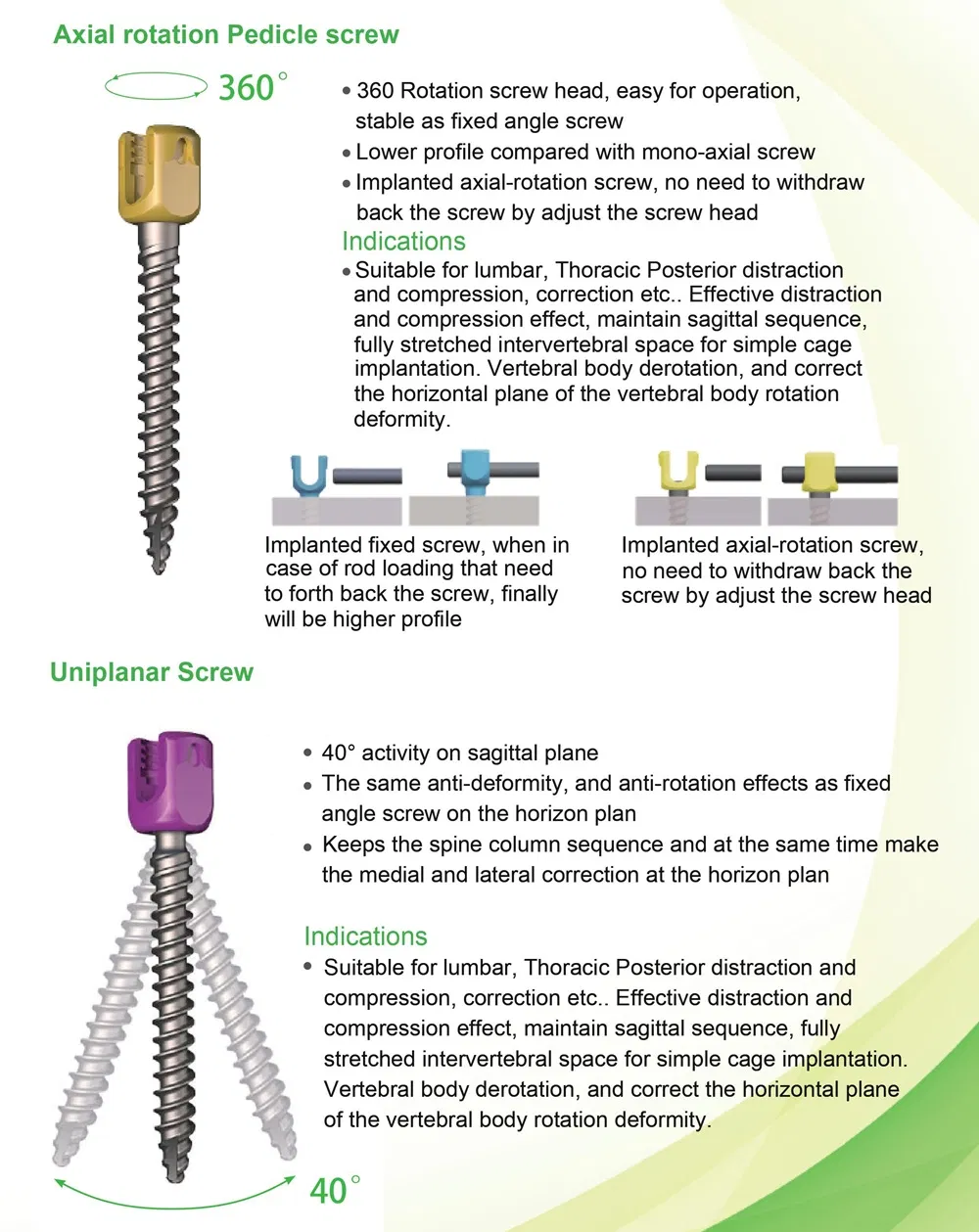
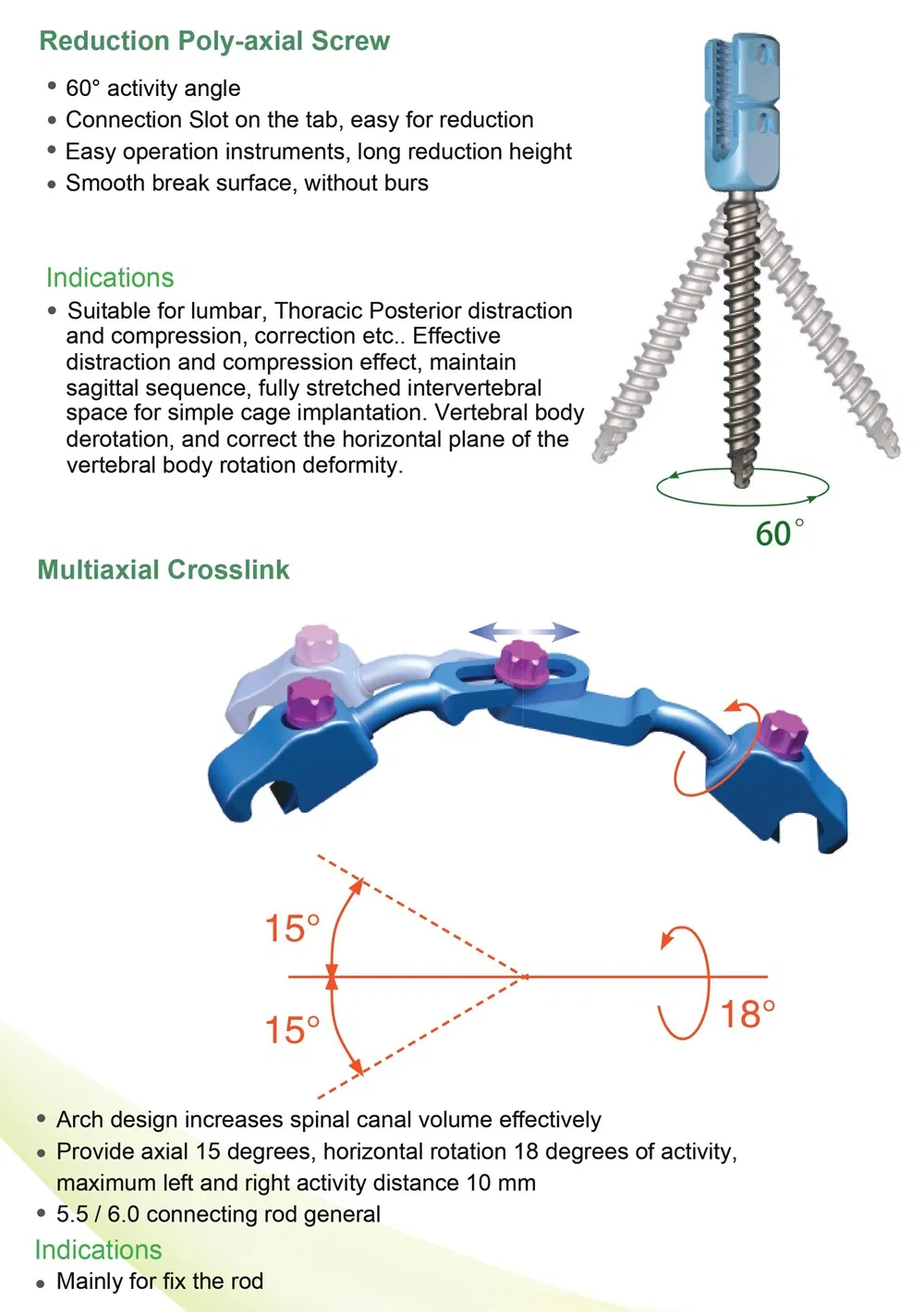
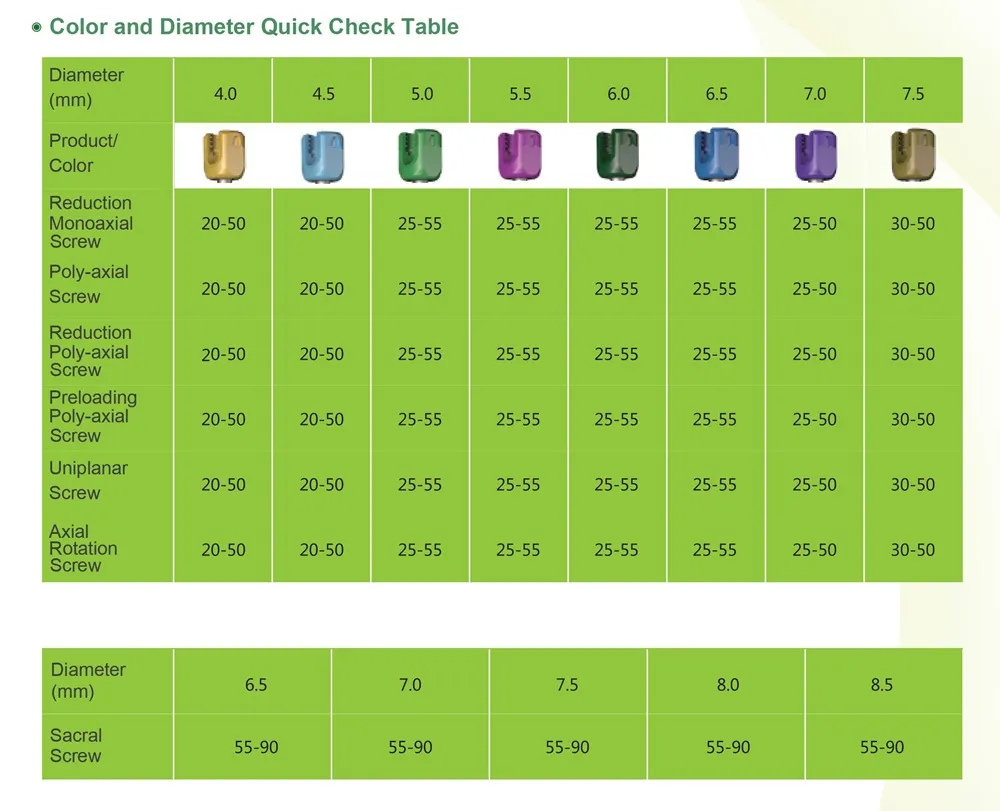


- Implants: Inner plastic bag + outer carton box.
- Instruments Set: Inner instruments box + outer carton box.
- Customized packing design service available.
- By air, DHL, UPS, FedEx, etc.



