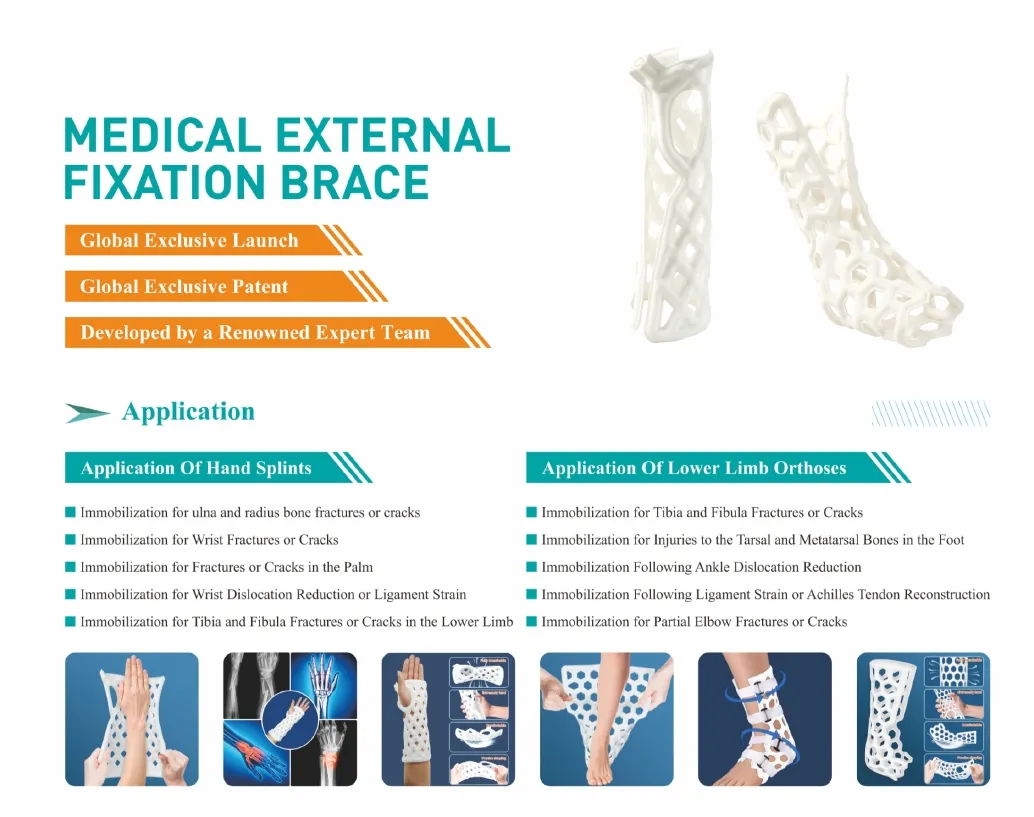
Portable Hand External Fixation System for Trauma and Orthopedic Surgery
◈ Immobilization for ulna and radius bone fractures or cracks
◈ Immobilization for Wrist Fractures or Cracks
◈ Immobilization for Fractures or Cracks in the Palm
◈ Immobilization for Wrist Dislocation Reduction or Ligament Strain
◈ Immobilization for Tibia and Fibula Fractures or Cracks in the Lower Limb
Frequently Asked Questions
1. What is the primary use of this orthopedic splint?
It is designed for the immobilization of fractures or cracks in the ulna, radius, wrist, palm, and lower limb bones like the tibia and fibula.
2. How is the product sterilized for safety?
The splints undergo Ethylene Oxide (EO) sterilization to ensure they are non-toxic, pyrogen-free, and safe for clinical use.
3. What certifications does the external fixation system have?
This product is certified under CE, ISO13485, and GMP standards, meeting international safety and quality requirements for Class II medical instruments.
4. What is the shelf life and storage recommendation?
The product has a shelf life of two years. It comes in a sterile blister package and should be stored according to international safety standards.
5. Is it possible to request samples before a bulk order?
Yes, samples are available upon request. The minimum order quantity (MOQ) starts from a single unit.
6. Does the product support logo customization?
Yes, we provide logo printing services and support both brand name and OEM requirements.




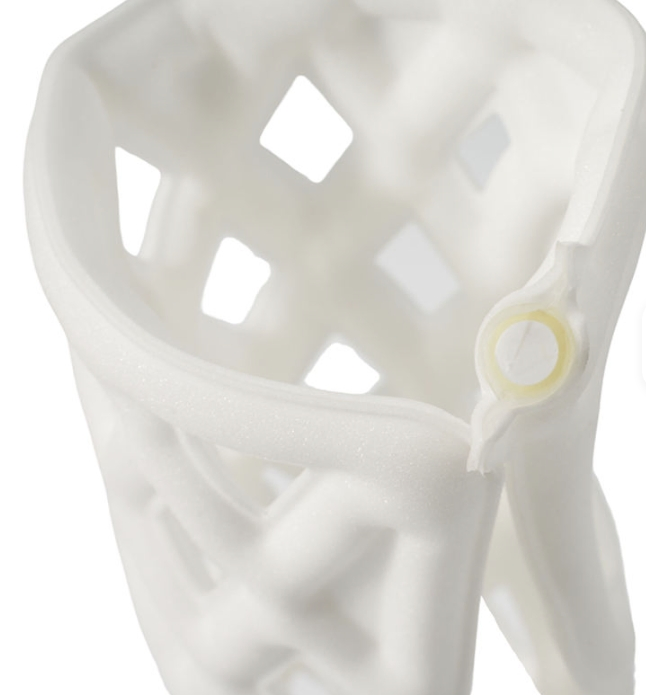
 Medisage Medical
Medisage Medical