 Medisage Medical
Medisage Medical










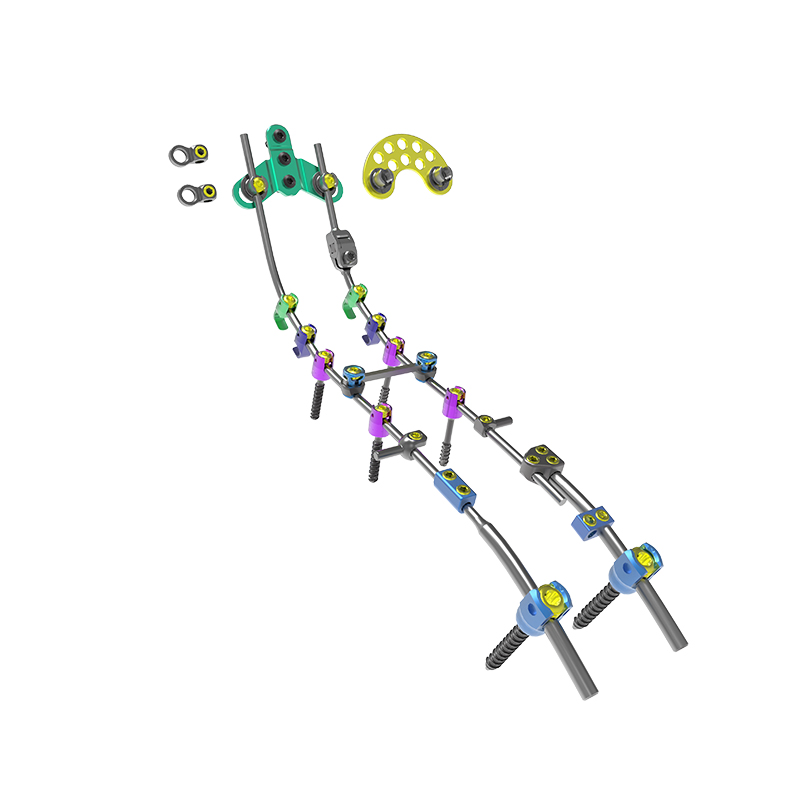
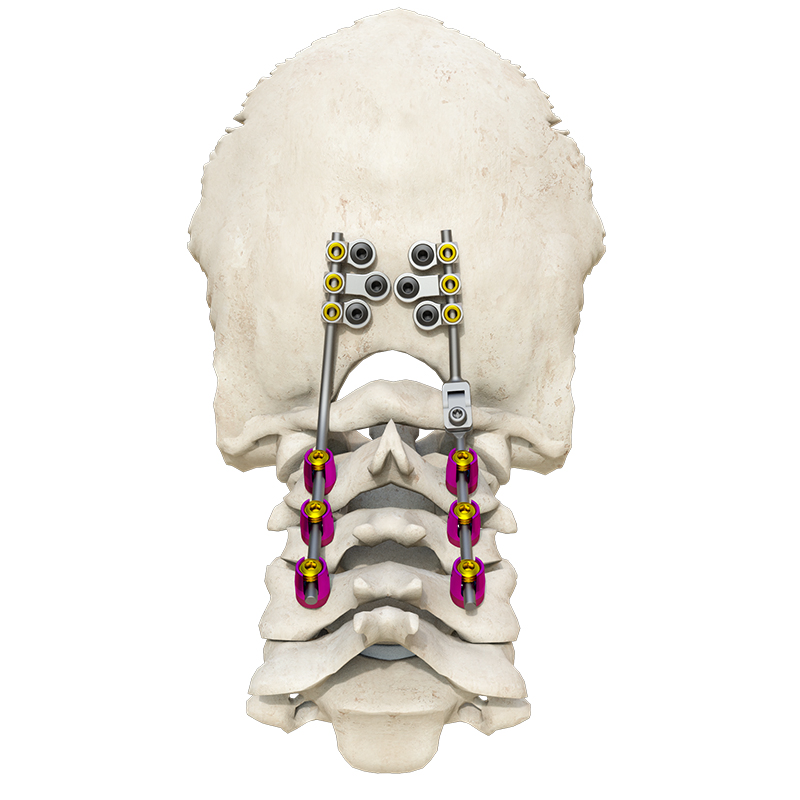
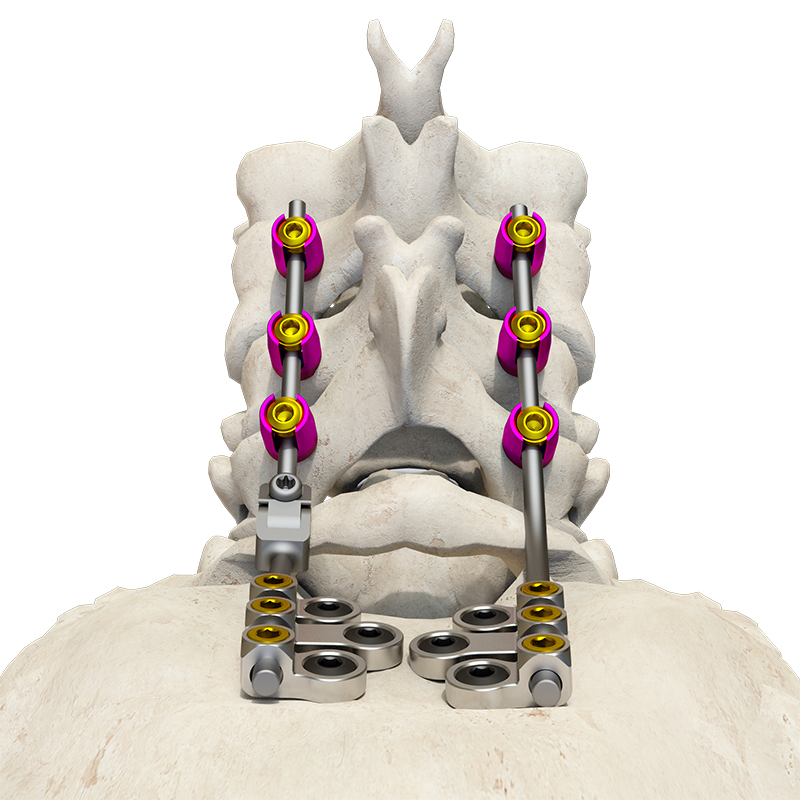
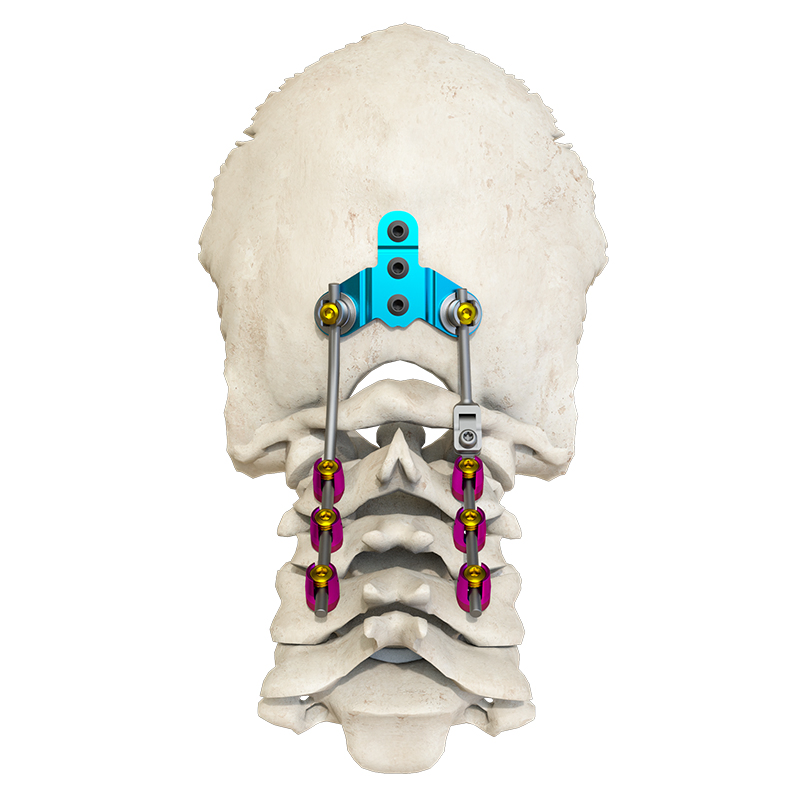
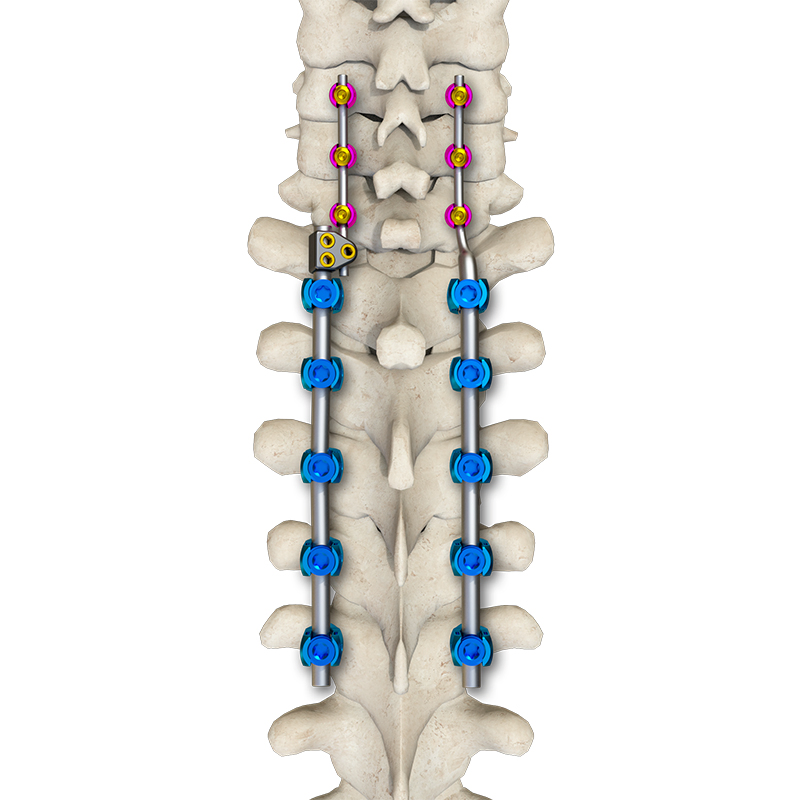
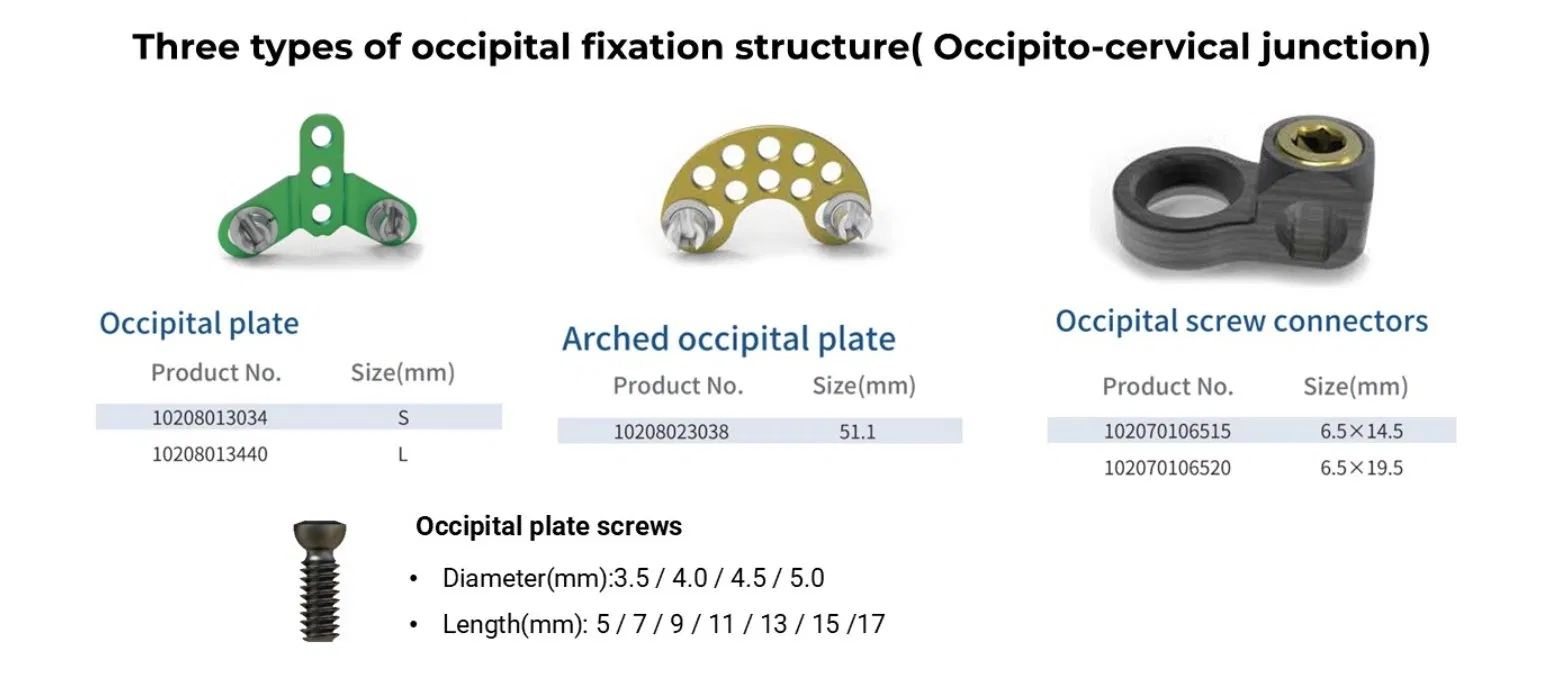
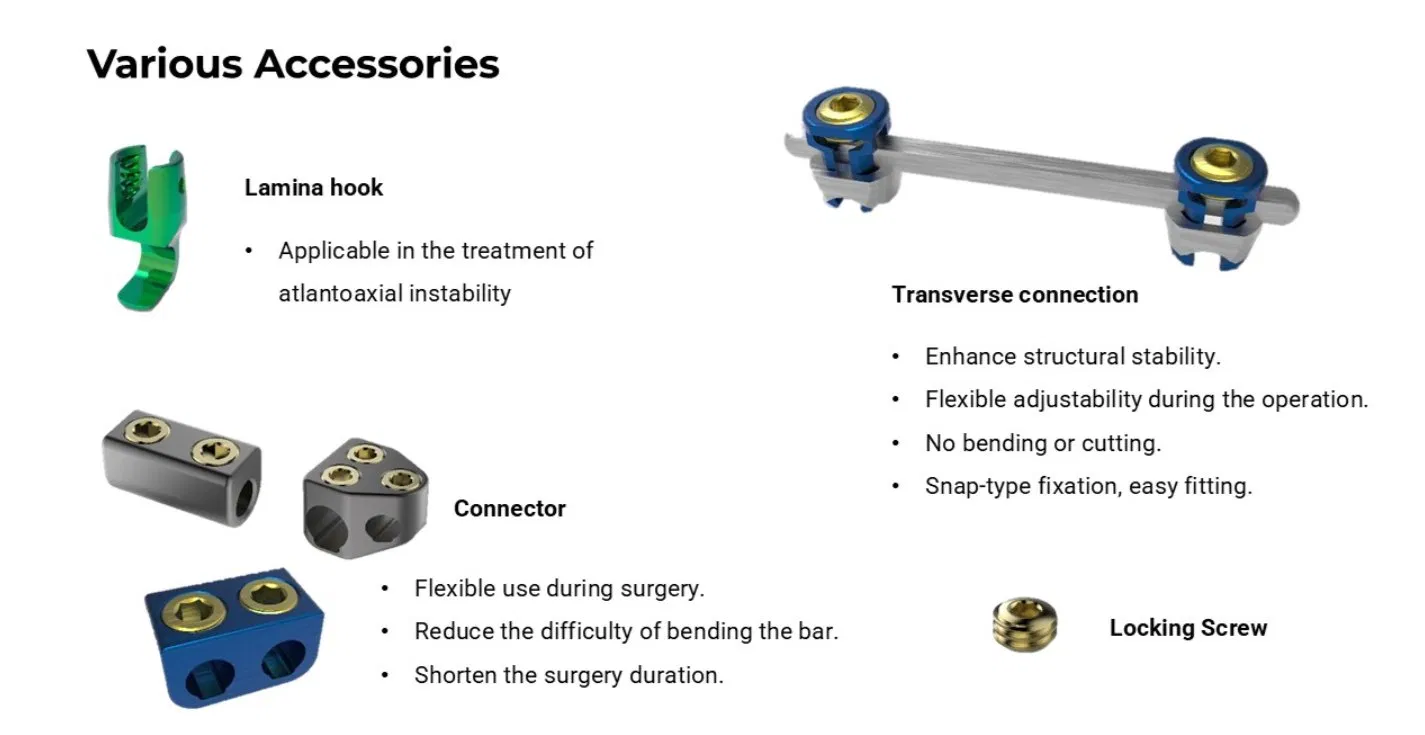
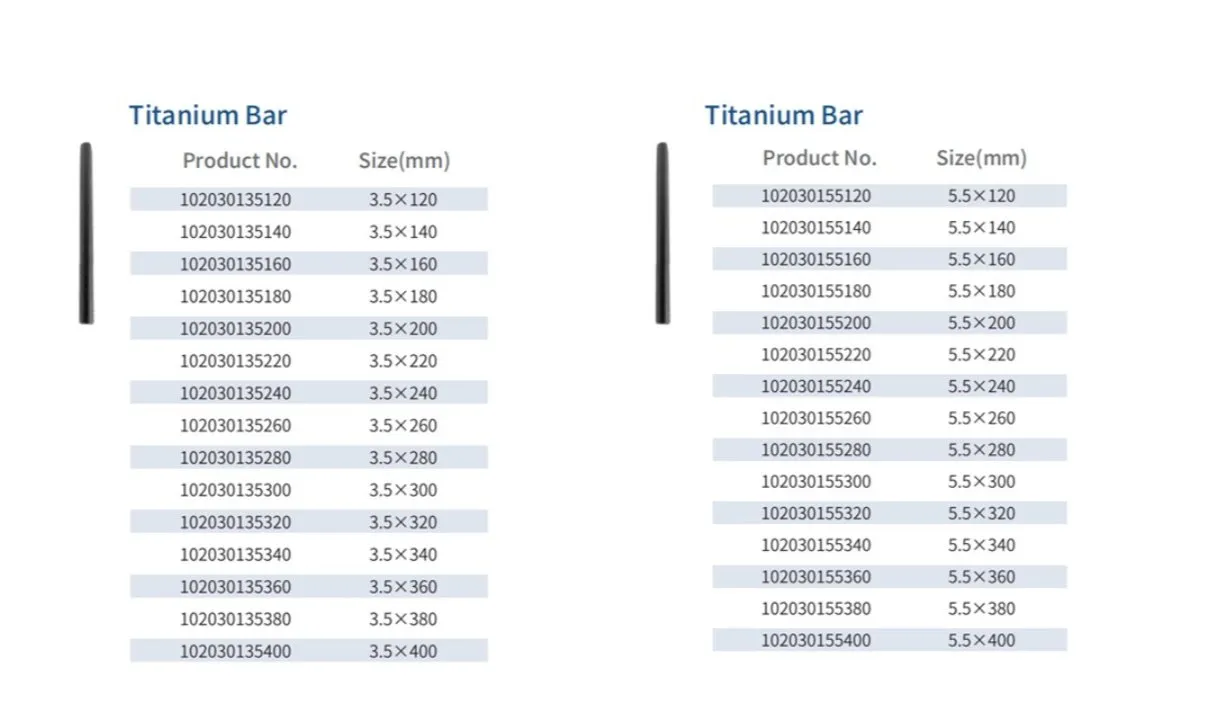
The Posterior Occipital-Cervical-Thoracic Spinal System is engineered for posterior spinal fixation and fusion from the occiput through to the thoracic spine. This sophisticated system integrates polyaxial screws, titanium rods (available in standard, dual-diameter, and adjustable configurations), crosslinks, lateral connectors, axial and domino connectors, occipital screws, plates, and lamina hooks.
Material: All components are manufactured from high-grade TC4 titanium alloy for superior biocompatibility and strength.








A Leading Manufacturer of Standardized Orthopedic Implants
We provide a comprehensive range of orthopedic solutions including trauma, spine, joints, sports medicine, and CMF products, accompanied by full surgical instrument sets. Designed for high-volume distribution, our products feature low MOQs and rapid turnaround times.
Our infrastructure includes in-house R&D, certified laboratories, and modern production lines, supporting both standardized supply and customized OEM/ODM services for global partners.